| |
Photodynamic Therapy
with Porphyrins
Copyright
© 1995–97
by Mike P. Gagel, RN, MSN
Overview of
Photodynamic Therapy
PHOTODYNAMIC THERAPY (PDT) is
the combination of light and light sensitive agents (such
as porphyrins) in an oxygen-rich environment. Porphyrins
are a component of hemoglobin, which in turn is a
component of red blood cells. Hemoglobin is what carries
oxygen in the blood. When porphyrins are not used as a
component of hemoglobin, they can absorb energy from
photons (particles of light) and transfer this energy to
surrounding oxygen molecules. Toxic oxygen species such
as singlet oxygen and free radicals are thus formed.
These chemicals are very reactive and can damage
proteins, lipids, nucleic acids and other cellular
components. Just as chlorophyll in plants utilizes energy
from sunlight to produce sugar, porphyrins utilize energy
from light to produce toxic oxygen species.
Modern PDT originated
at the turn of the century in Germany. Researchers
experimenting with self-injection of porphyrins noted
sunburns due to PDT reactions in their skin. Derived from
animal hemoglobin, two forms of porphyrin are well known:
hematoporphyrin derivative (HPD) and porfimer sodium
(Photofrin®). Photofrin® is in Phase III clinical trials and has
received approval in Canada for use with bladder
carcinoma where treatment with BCG vaccine has failed.
(Photofrin® has been approved in other countries for
treatment of esophageal cancer and lung cancer.) These
first generation photosensitizers display prolonged and
generalized photosensitivity of the skin as their primary
side effect. Second generation photosensitizers exhibit
far less photosensitization and are now in early clinical
trials (one example is BPD verteporfin). BPD verteporfin
was recently in Phase I/II clinical trials for:
- primary skin carcinoma
- cutaneous lesions where cancer has metastasized
to the skin, and
- chronic stable plaque psoriasis.
We are currently evaluating BPD
verteporfin in Vancouver, BC, Canada for treatment of
chronic stable plaque psorasis in patients who also have
psoriatic arthritis.
Lasers are the
primary light source for activation of porphyrins because
laser light is monochromatic (exactly one colour),
coherent (light waves are parallel permitting precise
focusing), and intense (allowing for shorter treatment
times). Light Emitting Diodes (LEDs) and florescent light
sources are now being used as alternative light sources
as they are more convenient than lasers but do result in
longer treatment times.
Go
to: Clinical Trials, Typical PDT, Photofrin®, BPD, Vancouver Trials, QLT
Clinical Trials
Clinical trials are drug studies. There are four
phases of clinical trials:
- Phase I – laboratory and animal
studies leading to a few studies with human
volunteers to determine a drug's safety
(e.g., establish side effects, how the body uses
the drug, how the drug circulates, how it is
excreted).
- Phase II – once safety is
established, more testing is done to determine a
drug's effectiveness
(i.e., how best to use the drug).
- Phase III – comparison
with standard treatments.
- Phase IV – combination
with standard treatments (to determine if
superior results can be obtained).
Go
to: Overview PDT, Typical PDT, Photofrin®, BPD, Vancouver Trials, QLT
A typical PDT session
- Intravenous injection (I.V.) or topical
application of a photosynthesizing agent such as
a porphyrin.
- Permit time for systemic porphyrins (I.V.
injection) to be cleared from normal tissues and
be preferentially retained by rapidly growing
tissues (e.g., cancer or psoriasis), or for
topical porphyrins to be absorbed by the skin.
- Application of light to provide the catalyst for
chemical reactions.
- Generation of toxic oxygen species in illuminated
tissues.
- Tissue damage usually resulting from damage to
vasculature.
Go
to: Overview PDT, Photofrin®, BPD, Vancouver Trials, QLT
Photofrin®
- Manufactured by QLT
Phototherapeutics.
- In clinical trials for the past decade.
- First PDT agent to receive regulatory approval.
- I.V. injection and then light application within
a 48–72 hour period (to allow for clearing
of Photofrin® from normal tissue).
- Red light (630 nm wavelength) activation.
- Major side effect is generalized photosensitivity
lasting up to 6–8 weeks following I.V.
injection.
Go
to: Overview PDT, Typical PDT, BPD, Vancouver Trials, QLT
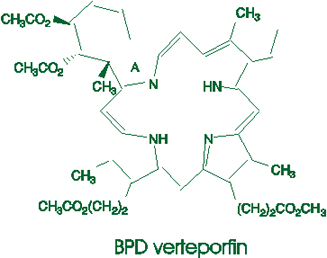
BPD verteporfin
- Manufactured by QLT
Phototherapeutics.
- In clinical trials since 1992.
- I.V. injection and then light application within
a 1.5–6 hour period (advantage over
Photofrin®).
- Activated by red or ultraviolet A (UVA) light.
- Red light (690 nm wavelength) activation
(advantage over Photofrin® when treating
cutaneous cancers as this wavelength of
light penetrates tissue more deeply than
630 nm light).
- UVA light (290–320 nm wavelength)
activation (able to use commonly
available light sources, e.g., Psoriasis
Clinic light booths).
- Major side effect is generalized photosensitivity
lasting up to 3–4 days (advantage over
Photofrin®).
- Photobleaching of residual BPD verteporfin in the
presence of normal light levels (Photofrin®
patients initially stay in darkened rooms).
- Promising results with skin cancer (, et
al. in press), and psoriasis
(Hruza, L., et al. in press).
Go
to: Overview PDT, Typical PDT, Photofrin®, Vancouver Trials, QLT
Vancouver Trials:
Our current Psoriasis/Psoriatic Arthritis Trial is
full (16Apr97). For more information please call:
RN, MSN
The Skin Care Centre
835 West 10th
Avenue
Vancouver, BC V5Z 4E8
CanadaTel: +1 (604) 875-5254
Fax: +1 (604) 873-9919
|
Click here for Q&As from our
Psoriasis Studies.
Go
to: Overview PDT, Typical PDT, Photofrin®, BPD, QLT
For information on
Photofrin® or BPD verteporfin, contact:
QLT Phototherapeutics Inc.
520 West 6th
Avenue
Vancouver, BC V5Z 4H5
CanadaTel: +1 (604) 872-7881
Tel: +1 (800) 663-5486
Fax: +1 (604) 875-0001
|
Go
to: Overview PDT, Typical PDT, Photofrin®, BPD, Vancouver Trials
Created:
06Dec95. Revised: 16Apr97.
Link
to information on Photodynamic
therapy of psoriasis
Return to Lions Laser Skin Centre
Homepage
Return to DermWeb
|
|